In a previous article, I explained how gases are exchanged at the respiratory membrane. Today, I will be talking about gas exchange at the tissues namely, the unloading of oxygen and loading of carbon dioxide onto red blood cells. This is a tricky concept at first to understand so don’t panic if it takes you a little longer to understand than the Bohr effect.
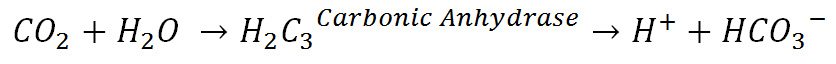
The Carbon dioxide that diffuses into bed blood cells forms H2CO3 (Carbonic acid) with the presence of carbonic anhydrase (an enzyme that increases the rate of formation of carbonic acid 5000-fold) which then dissociates into H+ ions and HCO3– ions. The HCO3– ions diffuse into the plasma while the H+ ions get buffered by haemoglobin. Carbaminohaemoglobin forms from this reaction.
Diffusion of carbon dioxide is driven by partial pressures; at the tissues, the PCO2 of arterial blood is 40mmHg, the PCO2 of the tissues is 46mmHg and the PCO2 of the interstitium and venous blood is 45mmHg.
One (1) mmHg of pressure doesn’t seem like much of a difference however, carbon dioxide is over 20 times more diffusable than oxygen and this small difference is plenty enough to create a pressure gradient.
Carbon dioxide in the blood is transported as 23% carboaminohaemoglobin, 7% CO2 and 70% H2CO3.
Offloading Carbon Dioxide at the Alveoli
Deoxyhaemoglobin, that is, haemoglobin without any oxygen bound to it, binds hydrogen ions more readily than oxygenated haemoglobin. This means that when oxygen is bound to haemoglobin its affinity for carbon dioxide is reduced. When oxygen binds to haemoglobin, it becomes a stronger acid, displacing carbon dioxide. This increase in acidity causes the haemoglobin to release more hydrogen ions which bind to bicarbonate ions creating carbon dioxide and water. This is called the Haldane Effect.
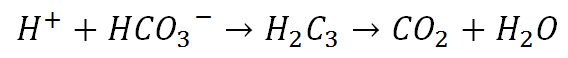
The resulting carbon dioxide diffuses out of the cells and is breathed out on expiration.
Part of the importance of this reaction is that a high amount of carbon dioxide causes pH to decrease, in other words, acidity increases. The body can only buffer so much acid so the body’s response to this is to drive the reaction in this direction by breathing out more carbon dioxide. This is a very fast response and changes can occur within several minutes, increasing pH to its normal level.
