Hemoglobin and myoglobin are two of the most important proteins in the body. They are a great example of the old biology maxim, structure equals function, and they show just how important proteins are to organisms. Their functions vary slightly, but they are vital for oxygen transport.
The Heme Group
The heme portion of myoglobin and hemoglobin is extremely important because it aids in oxygen binding. A heme group consists of a central iron atom (Fe II in this case) and it is surrounded by a porphyrin ring.
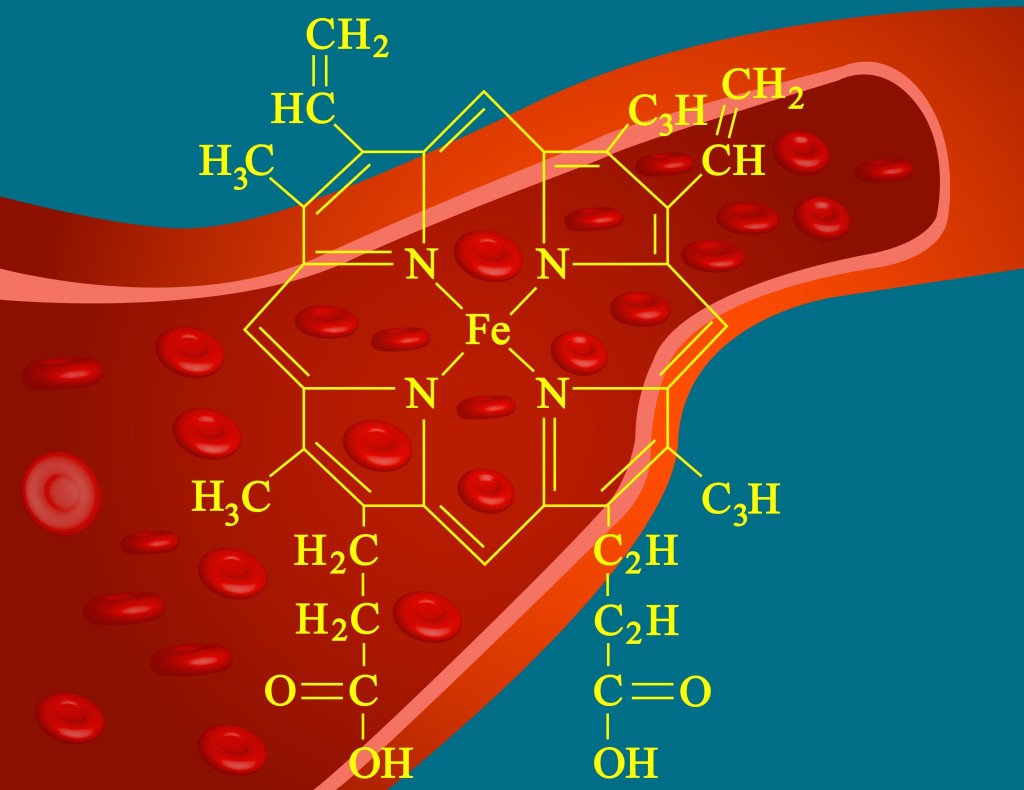
To put it simply, a porphyrin ring is a group of organic aromatic compounds. Heme acts as a cofactor (or prosthetic group) for hemoglobin and myoglobin.
A cofactor is not a protein, but it is important for the biological function of many proteins, including enzymes. The function of the heme group is to allow reversible binding of oxygen. Heme also provides the pigment for erythrocytes (red blood cells).
There are a few important amino acids to remember when discussing these proteins: valine, histidine and phenylalanine. The protein portions prevent the oxidation of Fe II to Fe III, which would inhibit the binding of oxygen.
Histidine serves an additional important function. Carbon monoxide has a much higher affinity for heme than oxygen. Histidine prevents the binding of carbon monoxide because CO cannot fit due to the bent conformation. Oxygen does bind in a bent fashion, and can fit with histidine.
Myoglobin
Myoglobin is the simpler of the two proteins, at least as far as structure goes. It is responsible for the storage of oxygen.
Myoglobin exhibits secondary structure, which is the folding of the linear chain of amino acids that make up the protein. Myoglobin has one subunit, which consists of α-helices (resemble spiral staircases) and β-sheets. The structure is stabilized by hydrogen bonds.
Ligands (molecules that bind to a central metal atom) bind independently to myoglobin, which means that the binding of a molecule has no influence on the binding of another. In the presence of low partial pressure, less oxygen will bind to myoglobin. More oxygen will bind when the partial pressure is higher. Myoglobin is less efficient at delivering oxygen and is rarely found outside of tissues.
Hemoglobin
Hemoglobin exhibits quaternary structure, which describes the folding of a protein that has multiple subunits. The function of hemoglobin is to transport oxygen to the tissues. Hemoglobin has four subunits, which means there are 4 proteins that fold to form a larger protein complex.
Like the subunit of myoglobin, the structure consists of alpha-helices stabilized by hydrogen bonds. Unlike myoglobin, hemoglobin has two conformations. The first conformation is the T-state, or deoxyhemoglobin. In this state, hemoglobin can be described as “tense” and has a low affinity for oxygen. When deoxygenated, hemoglobin appears blue. If oxygen binds to hemoglobin in this state, it becomes unstable and will shift to the R-state if enough oxygen binds to the protein.
The R-state (oxyhemoglobin) is the exact opposite. It is “relaxed” and has a high affinity for oxygen, meaning that it wants to bind with oxygen in order to become more stable. As a result of being oxygenated, the “R” state is red in color. Hemoglobin is an allosteric protein, meaning that the binding of oxygen at one site will influence the binding at another site.
Oxygen Transport Made Easy
Hemoglobin and myoglobin work together for efficient transport of oxygen from the lungs to the tissues. This can be explained in a few simple steps.

First, oxygen is inhaled at a high pressure in the lungs. High pressure facilitates the binding of oxygen to hemoglobin and allows the protein to hold onto it.
Hemoglobin transports the oxygen to areas with lower pressure, such as respiring tissue. Oxygen separates from the hemoglobin at lower pressure and oxidizes metabolic fuels, such as carbon dioxide. After this separation, oxygen binds to myoglobin. Finally, the oxygen and carbon dioxide are returned to the lungs, where the carbon dioxide is exhaled out of the body.
Hemoglobin and myoglobin are two proteins that are found in organisms that are crucial for their survival. These proteins have similar functions, but their individual structures make them different. As a result, hemoglobin and myoglobin are great examples of how structure equals function.

Hi 🙂
Thanks for the article. But shouldn’t hemoglobin be the protein of transport and myoglobin serve to store oxygen in muscle?
very clearly explained. good overview
Dr Densil ,kristen,you are a good explaner.explaned good artcle.have agood day
I have a doubt in ur structuraral explation for the hemoglobin, I think bottom position of the structure may be wrong. Bz the valancy of d carbon atom on the side chain is not complete