Proteins are among the most important, and intriguing, molecules in the body. There is a wide array of proteins and functions. Their structure is just as complex and ornate, not unlike a piece of brilliant art. They exist in all shapes and sizes, and without them, our bodies would cease to function. Proteins are very complex and there is much to discuss, but this article will cover the most essential, basic facts.
Protein Function
Proteins have a wide array of crucial functions in our bodies. They store amino acids, function as antibodies, act as hormones, have structural functions, transport important molecules and last but certainly not least, proteins can act as enzymes.
There are many examples of each type, but everyone is familiar with at least a few of them. Some of the most common include transport proteins, enzymes, hormones and structural proteins. Hemoglobin and myoglobin are examples of transport proteins and are responsible for oxygen transport, while pepsin is an enzyme that is essential for the digestion of food.
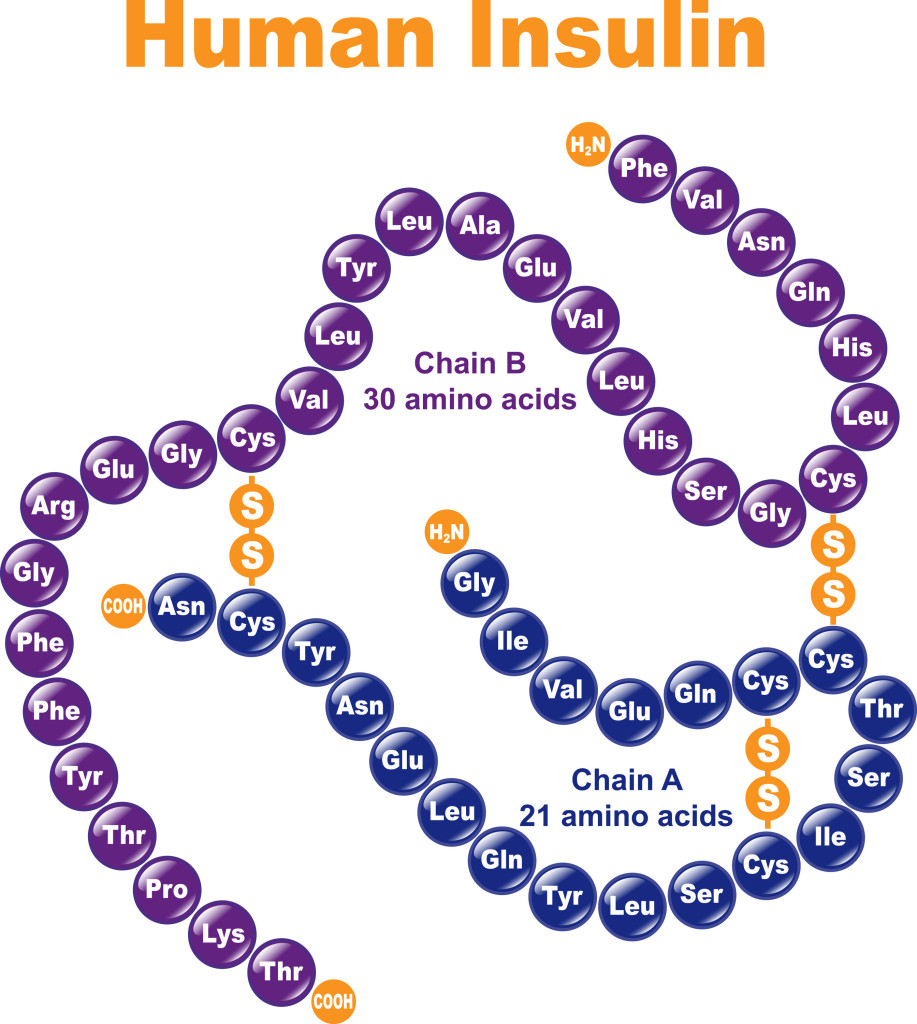
Hormones, such as insulin, are proteins that maintain essential bodily functions. Structural proteins help hold us together. These include connective tissues such as keratin and collagen. Of course, a protein’s function and structure go hand in hand.
Protein Structure
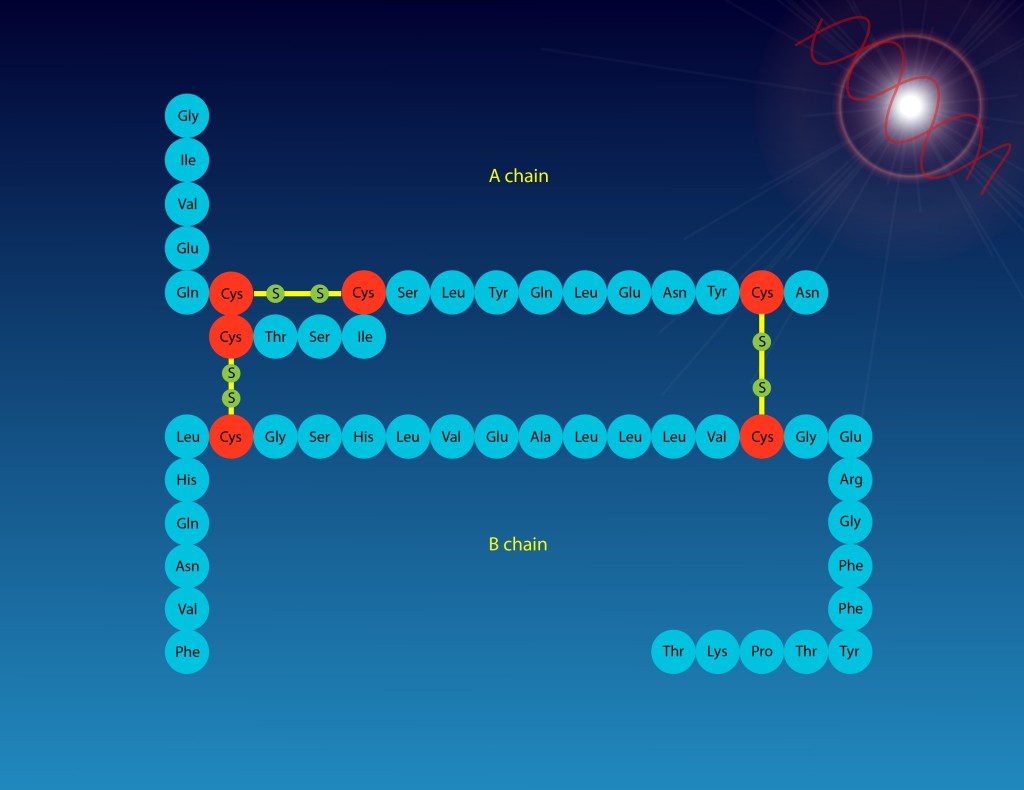
Amino acids are the building blocks of proteins and they are linked by peptide bonds to form a protein, or polypeptide. Amino acids consist of a carboxylic acid (COOH), an amine group (NH2), a carbon and a variable R group.
The simplest type of protein structure is called primary structure. This is simply the linear chain of amino acids that make up a protein.
Secondary structure is a bit more complex and refers to the arrangement or conformation of the amino acids. There are two main types of secondary structures: the α-helix and β-sheet. α-helices are right handed, resemble spiral staircases and they are stabilized by hydrogen bonds. Certain types of amino acids are much more common in α-helices than others. Alanine is a small amino acid and is more often found in α-helices. Glycine can also be found, but is less common. In this case, R groups are external while the hydrogen bonds are internal.
Bulkier amino acids, such as proline, are never found and would destabilize the α-helix. β-sheets come in two forms: parallel and anti-parallel. Parallel sheets occur when neighboring chains run in the same direction, and they run in the opposite direction in anti-parallel β-sheets. Instead of the R groups being external, they alternate in β-sheets. Small amino acids are common, while large amino acids destabilize β-sheets. Parallel β-sheets are less stable because they hydrogen bonds are distorted instead of linear, like in antiparallel β-sheets.
Tertiary structure is the folding of secondary structures and is the 3D structure of the protein that we see in pictures. The stabilization of this structure is more complex than the previous types. Hydrogen bonds, hydrophobic interactions, ionic bonds, and disulfide bonds are involved in the stability of tertiary structures.
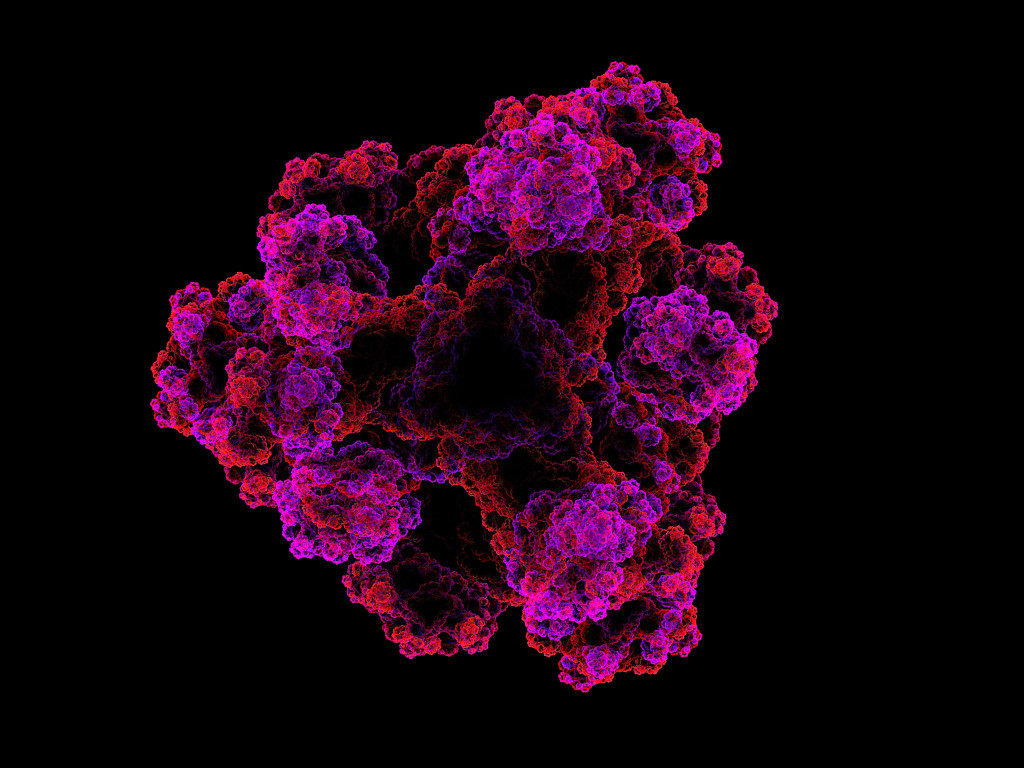
There are four types of tertiary structures. The most common is the β-α-β motif in which the α-helix connects two β-sheets. Next is the β-hairpin, which is also common and consists of antiparallel β-sheets that are connected by tight turns. The α-α motif is the most favorable and is formed by tightly packed α-helices. The final type is the Greek key motif, which is created by the folding of β-hairpins to form four anti-parallel sheets.
Quaternary structures exist in proteins with multiple subunits and describe the folding and arrangement of these subunits. An example of a protein that exhibits quaternary structure is hemoglobin. The structure of proteins is vital, and can be maintained in a few different ways.
Protein Stability
A protein is only useful if it can remain stable. Otherwise, it will denature (fall apart) and the protein will no longer be functional. The environment the protein is in is crucial. The pH of the environment is the first way to maintain protein stability. If the environment is too acidic, the protein will denature. The same is also true for temperature. Cooler is better, especially for long term storage of proteins in the lab.
Proteases also maintain protein stability. Proteases are enzymes that cleave peptide bonds that connect the amino acids. The final way to stabilize a protein is the ability to adsorb surfaces. As you can see, the environment in our bodies and in the lab must be optimal for our proteins to carry out their various tasks.
Proteins are a complex group of molecules and this article is just the tip of the iceberg. They have a wide variety functions and forms. If there is a deficiency of any protein, our bodies can be negatively impacted.

Author is not really into her job.she hasnt been able to explain it,just kind of narrated whatever she read…
Hi Mansoor, different articles have different purposes. Kristen did indeed accomplish her goal for this article, which is to shed light on the structure and function of the proteins. I’m proud of the work she has done and is continuing to do. If the article doesn’t resonate with you, that’s fine. We have many others on topics you might be interested in.
All the best!
Thanks so much because i can understand easily